Success the First Time
Success the First Time
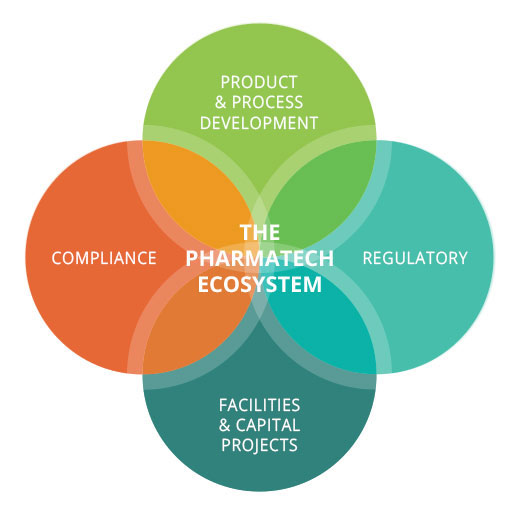
The drug and medical device development process often involves starts, stops, delays and do-overs. Success the first time requires an experienced team of development specialists who can provide a clear and proven roadmap. Staying on schedule requires the perfect mix of technical knowledge, practical business experience and broad regulatory compliance understanding – all supported by a culture of precise execution.
The drug and medical device development process often involves starts, stops, delays and do-overs. Success the first time requires an experienced team of development specialists who can provide a clear and proven roadmap. Staying on schedule requires the perfect mix of technical knowledge, practical business experience and broad regulatory compliance understanding – all supported by a culture of precise execution.